🧪 Maxwell–Boltzmann Distribution: Understanding Molecular Energy in Chemical Reactions
The Maxwell–Boltzmann distribution is a fundamental concept in chemical kinetics that explains how molecular energies are distributed within a system. Instead of all molecules having the same energy, molecules in a gas or solution exist across a range of energy levels. This distribution determines how many molecules possess enough energy to overcome the activation energy barrier required for a reaction to occur. Understanding this concept is crucial for MCAT chemistry, as it connects temperature, reaction rates, and activation energy in many chemical processes.
🧠 What Is the Maxwell–Boltzmann Distribution?
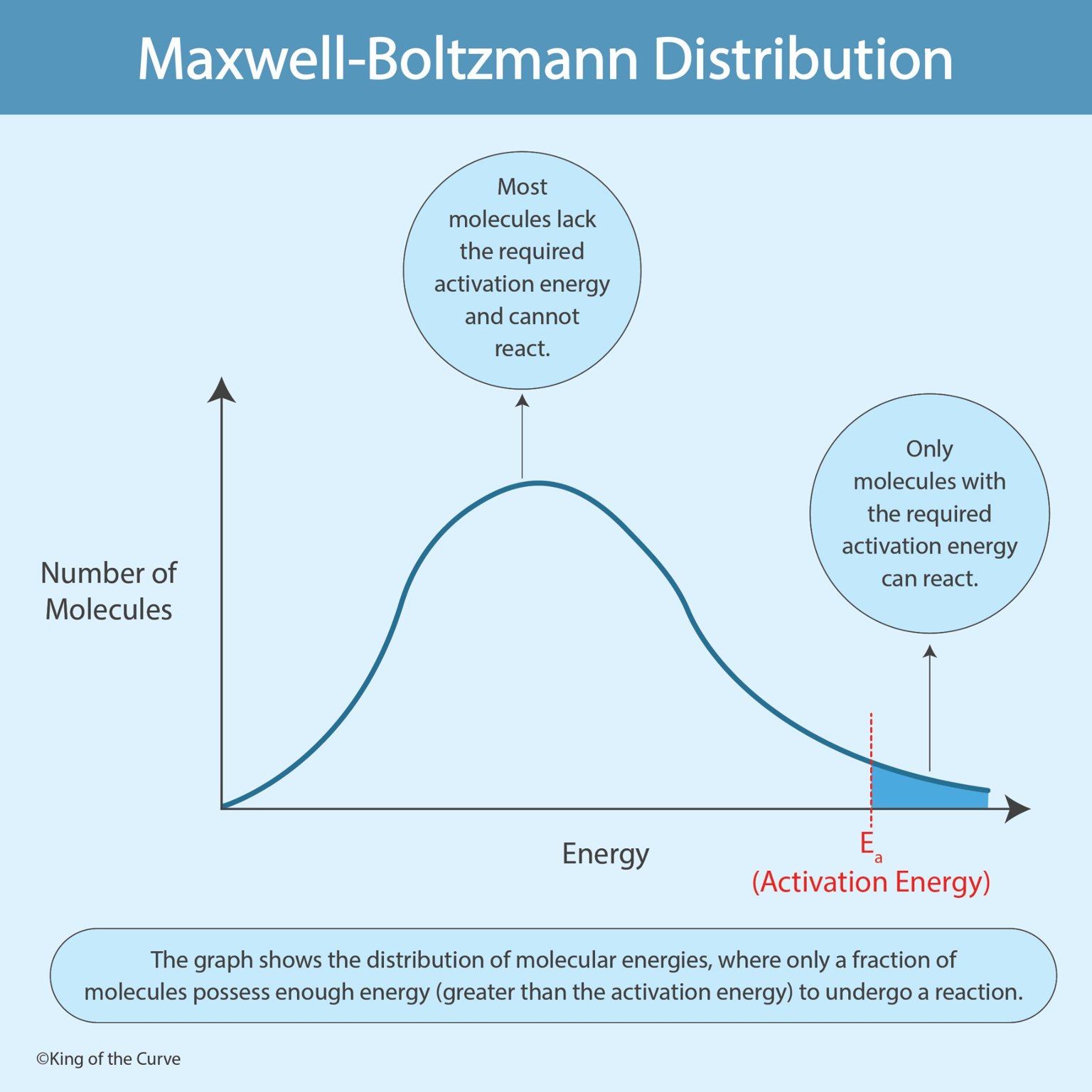
The Maxwell–Boltzmann distribution describes the statistical spread of molecular energies within a collection of particles. When plotted on a graph, the horizontal axis represents energy, while the vertical axis represents the number of molecules with that energy. Most molecules cluster around moderate energy levels, while fewer molecules exist at extremely low or extremely high energies.
⚡ Activation Energy and Chemical Reactions
For a chemical reaction to occur, molecules must collide with sufficient energy to overcome the activation energy (Ea) barrier. Only molecules with energy greater than this threshold can successfully react. On the Maxwell–Boltzmann curve, this is represented by the area to the right of the activation energy line, which shows the fraction of molecules capable of participating in the reaction.
🌡️ How Temperature Affects the Distribution
Temperature has a major effect on the shape of the Maxwell–Boltzmann distribution curve. As temperature increases, the curve becomes broader and shifts toward higher energy levels. This means more molecules gain enough energy to surpass the activation energy barrier, leading to faster reaction rates.
📈 Reaction Rate and Molecular Energy
Reaction rates depend on the number of effective molecular collisions. Since only molecules above the activation energy can react, the Maxwell–Boltzmann distribution helps explain why higher temperatures increase reaction speed. With more molecules reaching the necessary energy threshold, the probability of successful collisions increases significantly.
🧪 Catalysts and Activation Energy
Catalysts affect reactions by lowering the activation energy barrier rather than increasing the energy of molecules. On a Maxwell–Boltzmann graph, this effectively shifts the activation energy line to the left. As a result, a larger fraction of molecules possess sufficient energy to react, which increases the reaction rate without changing the overall energy distribution.
📊 Key Factors Affecting Reaction Rates
| Factor | Effect on Distribution | Result |
|---|---|---|
| Temperature increase | Curve shifts right and flattens | More molecules exceed activation energy |
| Catalyst presence | Lowers activation energy threshold | Larger fraction of molecules can react |
| Lower temperature | Curve shifts left | Fewer molecules have sufficient energy |
📚 MCAT Strategy: Interpreting Distribution Graphs
On the MCAT, Maxwell–Boltzmann graphs often appear in questions involving reaction kinetics, temperature effects, and catalysts. Students may be asked to determine how the curve changes when temperature increases or when a catalyst is introduced. Recognizing that the area beyond the activation energy represents reacting molecules helps quickly interpret these diagrams.
📈 Learning Chemical Kinetics with Visual Tools
Visualizing molecular energy distributions makes kinetic concepts easier to understand. King of the Curve provides hundreds of visual explanations for key MCAT chemistry topics, helping students grasp complex ideas like reaction kinetics, activation energy, and energy distributions. Explore more visual study tools at mcat.kingofthecurve.org, a leading resource for MCAT concept visuals and science learning support.
Frequently Asked Questions (FAQs)
-
Aim for 4-6 focused hours, ensuring you incorporate breaks to avoid burnout.
-
Practice mindfulness techniques, take practice exams under realistic conditions, and maintain a balanced lifestyle.
-
Set short-term goals, seek support from mentors, and reward yourself for small achievements.
-
Regular exercise improves focus, reduces stress, and enhances overall mental clarity.
-
KOTC offers personalized learning tools, gamification features, and adaptive question banks to help students stay on track without burnout.