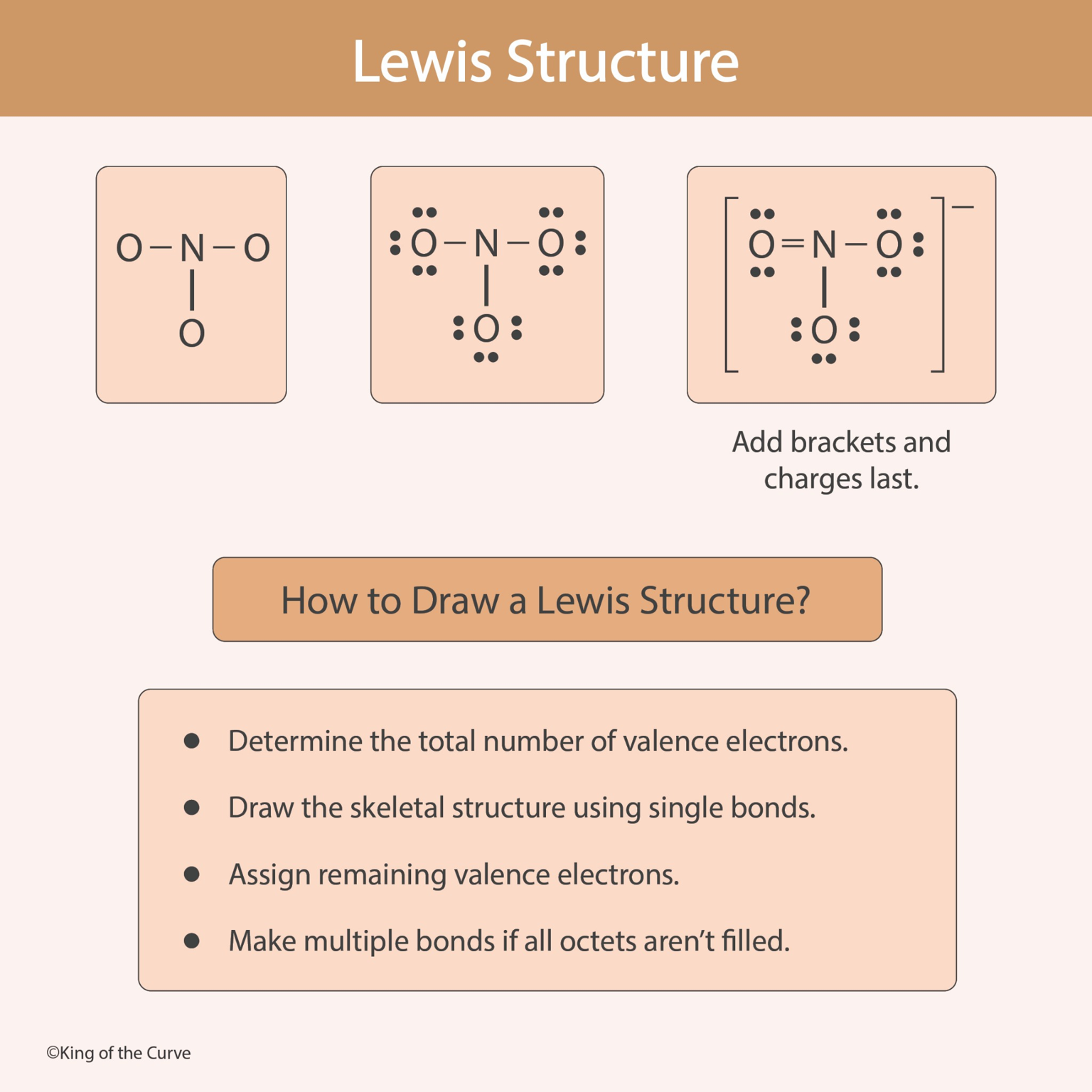
🧪 Lewis Structures: A Step-by-Step Guide to Understanding Molecular Bonding
Lewis structures are one of the most fundamental tools used in chemistry to visualize how atoms share electrons to form molecules. These diagrams help illustrate bonding patterns, lone pairs, and molecular charge distribution. For students preparing for the MCAT or studying general chemistry, understanding how to draw Lewis structures is essential because it helps explain molecular geometry, chemical reactivity, and resonance structures that frequently appear in exam questions and scientific passages.
🧠 What Is a Lewis Structure?
A Lewis structure is a diagram that represents the valence electrons of atoms in a molecule. Valence electrons are the outermost electrons involved in chemical bonding. By arranging these electrons as dots around atomic symbols and connecting atoms with lines that represent shared electron pairs, chemists can predict how atoms bond and how molecules behave.
⚛️ Why Lewis Structures Matter in Chemistry
Lewis structures provide insight into bond formation, electron distribution, and molecular stability. They allow scientists to determine how many bonds an atom forms, where lone pairs are located, and whether a molecule follows the octet rule. This information is crucial for predicting molecular geometry, polarity, and chemical reactions.
✏️ Step 1: Determine Valence Electrons
The first step when drawing a Lewis structure is calculating the total number of valence electrons present in the molecule or ion. These electrons come from the outermost shell of each atom involved. For polyatomic ions, the total electron count must be adjusted based on the overall charge of the molecule.
🔗 Step 2: Draw the Skeletal Structure
Next, draw the basic framework of the molecule by connecting atoms with single bonds. Typically, the least electronegative atom is placed at the center, while hydrogen atoms and more electronegative atoms are positioned on the outside. Each bond represents a shared pair of electrons.
🧩 Step 3: Assign Remaining Electrons
After drawing the skeletal structure, distribute the remaining electrons as lone pairs around the outer atoms first, ensuring they satisfy the octet rule whenever possible. If electrons remain after filling the outer atoms, they are placed on the central atom.
📊 Summary of Lewis Structure Steps
| Step | Action | Purpose |
|---|---|---|
| 1 | Count total valence electrons | Determine available electrons |
| 2 | Draw skeletal structure | Establish bonding framework |
| 3 | Distribute remaining electrons | Complete octets for outer atoms |
| 4 | Form multiple bonds if needed | Achieve stable electron configuration |
🔄 Step 4: Create Multiple Bonds if Necessary
If the central atom does not have a complete octet after distributing electrons, electrons from surrounding atoms may be shared to form double or triple bonds. This step helps ensure that atoms achieve stable electron configurations whenever possible.
📚 MCAT Strategy: Recognizing Lewis Structures in Passages
On the MCAT, Lewis structures often appear in questions related to resonance, formal charge, molecular geometry, and chemical bonding. Students may be asked to determine which structure is most stable or predict molecular shape using electron distribution. Mastering the step-by-step process of drawing Lewis structures makes it much easier to analyze molecules presented in exam passages.
📈 Learning Chemistry with Visual Tools
Complex chemistry concepts become much easier to understand when paired with visual diagrams. King of the Curve provides hundreds of illustrated science explanations designed for MCAT preparation, helping students quickly grasp topics such as Lewis structures, molecular geometry, and bonding theory. Explore more visual study tools at mcat.kingofthecurve.org, a leading resource for MCAT concept visuals and science learning support.
Frequently Asked Questions (FAQs)
-
Aim for 4-6 focused hours, ensuring you incorporate breaks to avoid burnout.
-
Practice mindfulness techniques, take practice exams under realistic conditions, and maintain a balanced lifestyle.
-
Set short-term goals, seek support from mentors, and reward yourself for small achievements.
-
Regular exercise improves focus, reduces stress, and enhances overall mental clarity.
-
KOTC offers personalized learning tools, gamification features, and adaptive question banks to help students stay on track without burnout.