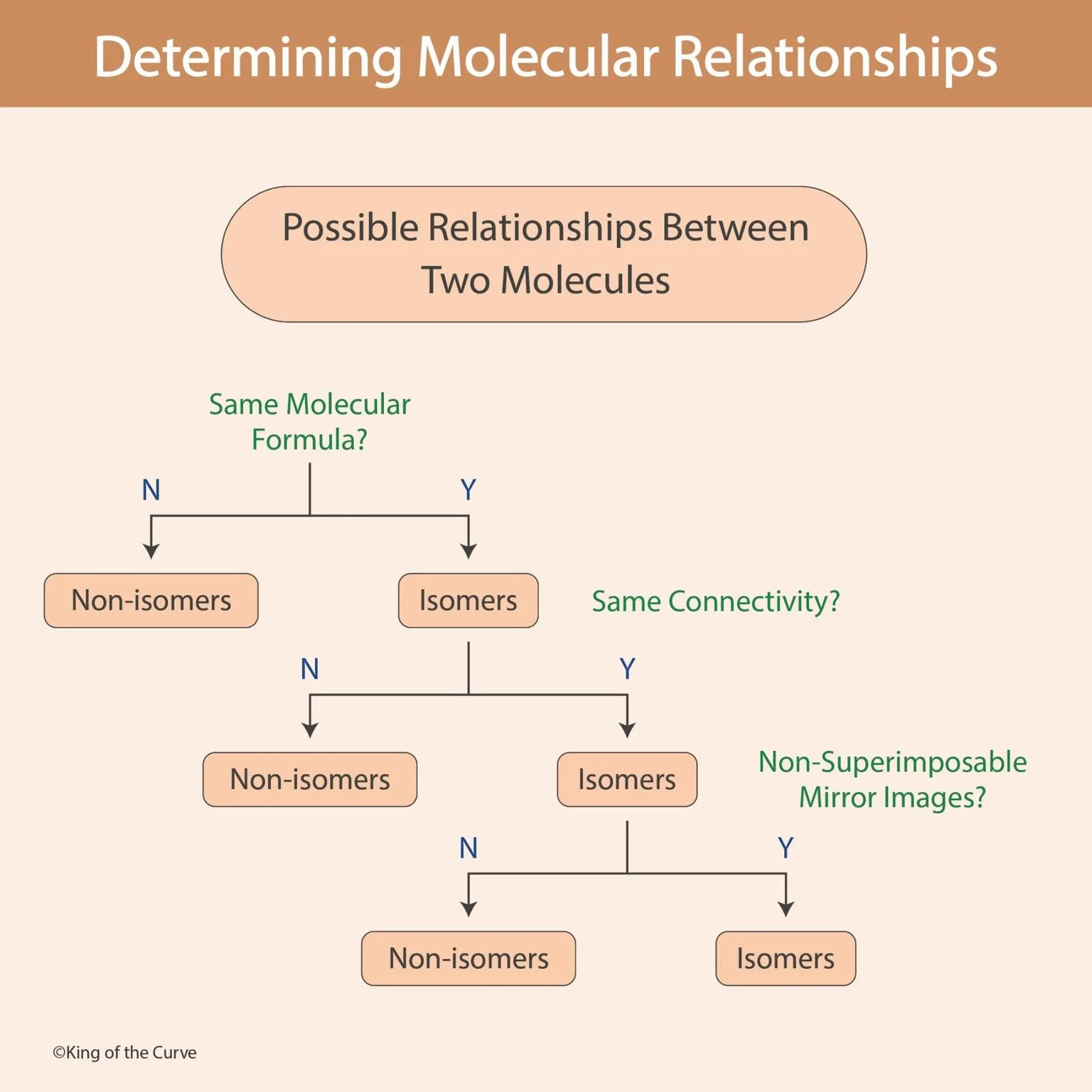
🧪 Determining Molecular Relationships
Understanding how molecules relate to one another is a fundamental concept in chemistry. Whether you're studying organic chemistry, biochemistry, or molecular biology, identifying whether two molecules are identical, isomers, or entirely different can help predict their properties, reactivity, and biological function. The diagram above provides a simple decision-making pathway to classify molecular relationships step by step.
🔍 Step 1: Do the Molecules Have the Same Molecular Formula?
The first question to ask is whether the two molecules share the same molecular formula. If they do not, they are classified as non-isomers, meaning they are entirely different compounds with different numbers or types of atoms.
If the answer is yes, the molecules are considered isomers. Isomers are compounds that have the same molecular formula but differ in structure or arrangement. This distinction is crucial because even small differences in structure can lead to significant changes in chemical and biological behavior.
🔗 Step 2: Do They Have the Same Connectivity?
Once molecules are identified as isomers, the next step is to determine whether they have the same connectivity—meaning the same atoms are bonded in the same order.
If connectivity differs, the molecules are structural (constitutional) isomers.
If connectivity is the same, the molecules are classified as stereoisomers.
This step helps distinguish whether differences arise from bonding patterns or spatial arrangement.
🪞 Step 3: Are They Non-Superimposable Mirror Images?
For molecules with the same connectivity, the final question focuses on their three-dimensional arrangement.
If yes, they are enantiomers (mirror images that cannot be superimposed).
If no, they are diastereomers (stereoisomers that are not mirror images).
This classification is especially important in biological systems, where molecular orientation can drastically affect function.
📊 Summary Table of Molecular Relationships
| Step | Question | Yes Outcome | No Outcome |
|---|---|---|---|
| 1 | Same molecular formula? | Isomers | Non-isomers |
| 2 | Same connectivity? | Stereoisomers | Structural isomers |
| 3 | Non-superimposable mirror images? | Enantiomers | Diastereomers |
⚗️ Real-World Applications
Understanding molecular relationships is critical in fields like pharmaceuticals, where different isomers of a drug can have drastically different effects. For example, one enantiomer of a compound may be therapeutically beneficial, while its mirror image could be inactive or even harmful. This is why drug design and testing place strong emphasis on stereochemistry.
In addition, industries such as food chemistry and materials science rely on molecular structure to determine taste, smell, and physical properties. Even slight variations in molecular arrangement can influence how substances interact with receptors or behave under different conditions.
🧠 Key Takeaway
By systematically asking three key questions about molecular formula, connectivity, and spatial arrangement you can accurately classify molecular relationships. This logical approach simplifies complex chemical analysis and provides a strong foundation for understanding structure-function relationships in chemistry.
Frequently Asked Questions (FAQs)
-
Aim for 4-6 focused hours, ensuring you incorporate breaks to avoid burnout.
-
Practice mindfulness techniques, take practice exams under realistic conditions, and maintain a balanced lifestyle.
-
Set short-term goals, seek support from mentors, and reward yourself for small achievements.
-
Regular exercise improves focus, reduces stress, and enhances overall mental clarity.
-
KOTC offers personalized learning tools, gamification features, and adaptive question banks to help students stay on track without burnout.