🧪 Drawing Lewis Structures: A Step-by-Step Guide for Beginners
Lewis structures are a fundamental tool in chemistry that help visualize how atoms bond and how electrons are distributed in a molecule or ion. By representing valence electrons as dots and bonds as lines, Lewis structures make it easier to understand molecular geometry, reactivity, and stability.
📘 What Are Lewis Structures?
Lewis structures (also called Lewis dot structures) show:
Valence electrons as dots
Covalent bonds as lines
Lone pairs as pairs of dots
They are especially useful for predicting:
Molecular shape
Bonding patterns
Formal charges
🧭 Step-by-Step: How to Draw Lewis Structures
Follow these steps systematically:
🔢 1. Find Total Valence Electrons
Add up valence electrons from all atoms
Add for negative charge
Subtract for positive charge
🏗️ 2. Draw the Skeleton Structure
Place least electronegative atom in the center
Connect atoms with single bonds
⚡ 3. Assign Remaining Electrons
Complete octets for outer atoms first
Then assign to central atom
🔗 4. Form Multiple Bonds if Needed
Convert lone pairs into double/triple bonds if octet is incomplete
➕ 5. Add Brackets and Charge
Required for ions
Write charge outside brackets
📊 Summary Table: Lewis Structure Steps
| Step | Action | Purpose |
|---|---|---|
| 1️⃣ Count Electrons | Calculate total valence electrons | Ensures correct electron distribution |
| 2️⃣ Skeleton | Draw basic structure with single bonds | Establishes atom connectivity |
| 3️⃣ Fill Octets | Assign electrons to complete octets | Stabilizes atoms |
| 4️⃣ Multiple Bonds | Create double/triple bonds if needed | Completes central atom octet |
| 5️⃣ Charge | Add brackets and charge | Represents ions correctly |
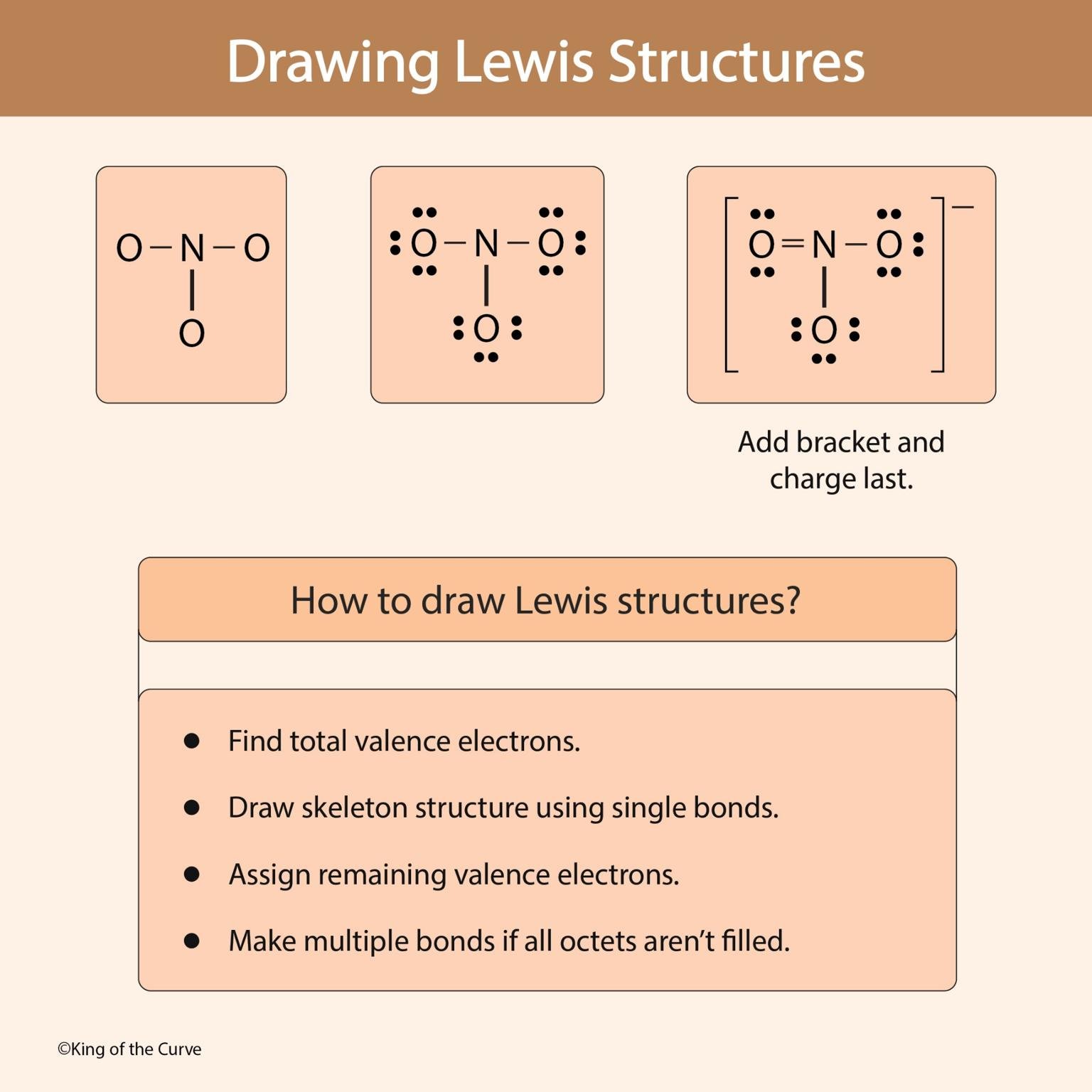
🧩 Example: Drawing a Polyatomic Ion
The image demonstrates:
Initial skeleton structure
Addition of lone pairs
Formation of a double bond
Final structure with brackets and charge
This stepwise approach ensures accuracy and stability.
⚠️ Common Mistakes to Avoid
❌ Incorrect electron counting
❌ Ignoring octet rule
❌ Forgetting formal charges
❌ Missing brackets for ions
🎯 Why Lewis Structures Matter
They help in:
Predicting molecular geometry
Understanding chemical bonding
Identifying resonance structures
Learning hybridization concepts
💡 Final Tips
Start with simple molecules
Always verify electron count
Minimize formal charges
Frequently Asked Questions (FAQs)
-
Aim for 4-6 focused hours, ensuring you incorporate breaks to avoid burnout.
-
Practice mindfulness techniques, take practice exams under realistic conditions, and maintain a balanced lifestyle.
-
Set short-term goals, seek support from mentors, and reward yourself for small achievements.
-
Regular exercise improves focus, reduces stress, and enhances overall mental clarity.
-
KOTC offers personalized learning tools, gamification features, and adaptive question banks to help students stay on track without burnout.