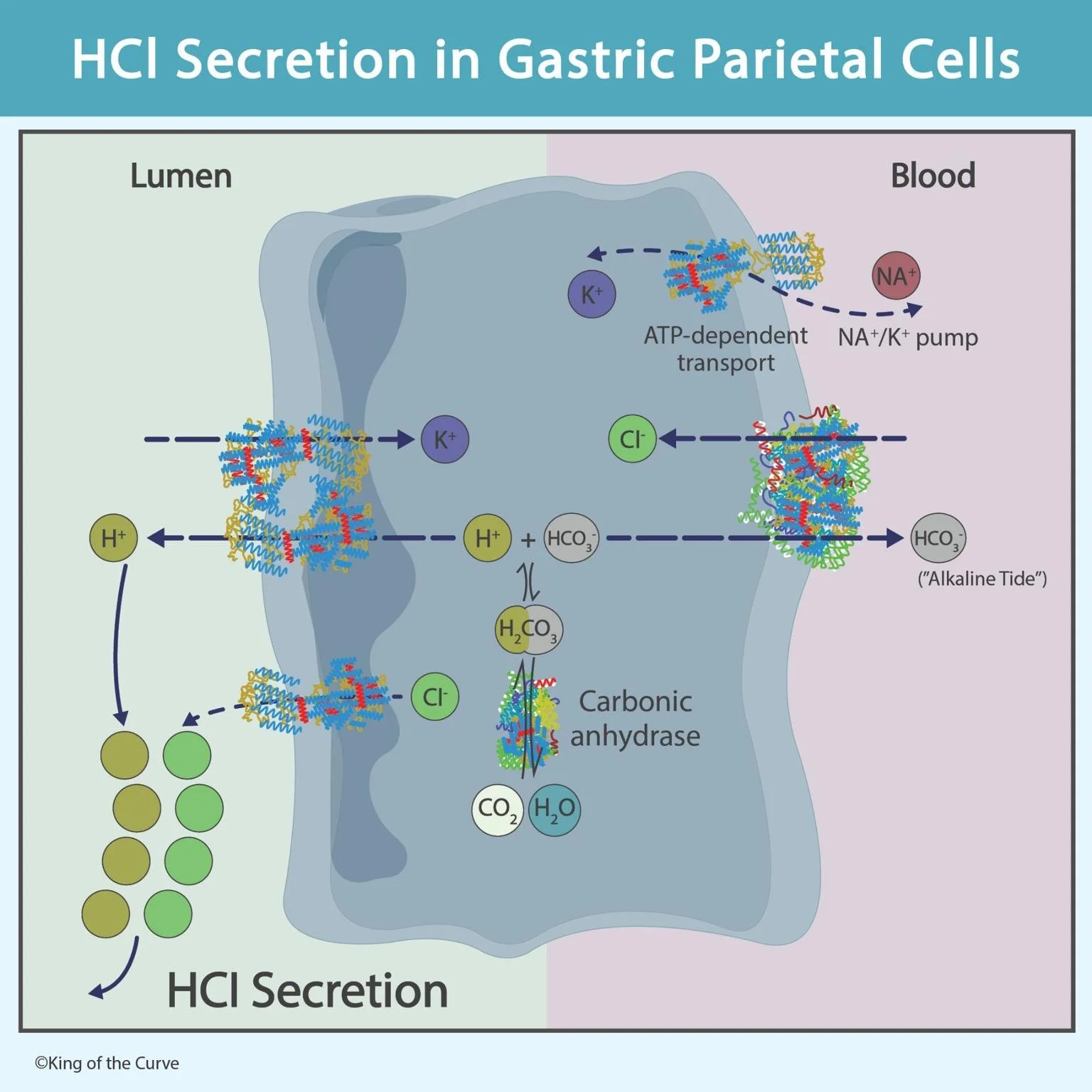
🧪 HCl Secretion in Gastric Parietal Cells: Mechanism Explained
Hydrochloric acid (HCl) secretion in the stomach is a vital physiological process that aids digestion, activates enzymes, and protects against pathogens. This process occurs in gastric parietal cells and involves a coordinated movement of ions across membranes.
🧬 Overview of Parietal Cell Function
Parietal cells are specialized epithelial cells located in the gastric glands of the stomach lining. Their primary role is to secrete:
Hydrochloric acid (HCl)
Intrinsic factor (important for Vitamin B₁₂ absorption)
⚙️ Step-by-Step Mechanism of HCl Secretion
1️⃣ Formation of Hydrogen Ions (H⁺)
Inside the parietal cell:
Carbon dioxide (CO₂) combines with water (H₂O)
This reaction is catalyzed by carbonic anhydrase
| Step | Reaction | Importance |
|---|---|---|
| 1. Hydration | CO2 + H2O → H2CO3 | Carbon dioxide combines with water to form carbonic acid. |
| 2. Dissociation | H2CO3 → H+ + HCO3− | Carbonic acid splits into hydrogen and bicarbonate ions. |
| Overall Reaction | CO2 + H2O → H2CO3 → H+ + HCO3− | Key reaction for bicarbonate handling and acid–base balance. |
👉 This produces:
Hydrogen ions (H⁺) → used to form acid
Bicarbonate ions (HCO₃⁻) → transported out
2️⃣ Proton Pump (H⁺/K⁺ ATPase)
H⁺ ions are actively transported into the gastric lumen
This occurs via the H⁺/K⁺ ATPase pump
Requires ATP (energy)
👉 Exchange:
H⁺ moves out
K⁺ moves in
3️⃣ Chloride Ion Movement (Cl⁻)
Cl⁻ enters the cell from blood via Cl⁻/HCO₃⁻ exchanger
Then diffuses into the lumen through Cl⁻ channels
4️⃣ Formation of HCl in the Lumen
In the lumen:
H⁺ + Cl⁻ → HCl (hydrochloric acid)
👉 This creates the highly acidic gastric environment (pH ~1–2)
5️⃣ Alkaline Tide (Bicarbonate Shift)
HCO₃⁻ produced earlier is transported into the blood
This causes a temporary rise in blood pH after meals
👉 Known as the “alkaline tide”
6️⃣ Role of Na⁺/K⁺ Pump
Maintains ionic balance
Pumps:
Na⁺ out
K⁺ into the cell
👉 Supports continuous function of the proton pump
🔁 Summary Table
| Component | Function |
|---|---|
| Carbonic anhydrase | Produces H+ and HCO3− |
| H+/K+ ATPase pump | Secretes H+ into the lumen |
| Cl− channel | Moves Cl− into the lumen |
| HCO3− exchanger | Sends bicarbonate to the blood |
| Na+/K+ pump | Maintains ion gradients |
| Lumen | Site of HCl formation |
🧠 Key Concepts to Remember
HCl is formed outside the cell (in lumen), not inside
Secretion is ATP-dependent
Alkaline tide occurs after meals
Proton pump inhibitors (PPIs) target the H⁺/K⁺ ATPase
💊 Clinical Relevance
Proton Pump Inhibitors (PPIs) like omeprazole block acid secretion
Overproduction → peptic ulcers, GERD
Underproduction → digestive issues, infections
📌 Final Takeaway
HCl secretion is a highly regulated, energy-dependent process involving ion exchange, enzymatic reactions, and membrane transport. Understanding this mechanism is essential for both physiology and pharmacology.
Frequently Asked Questions (FAQs)
-
Aim for 4-6 focused hours, ensuring you incorporate breaks to avoid burnout.
-
Practice mindfulness techniques, take practice exams under realistic conditions, and maintain a balanced lifestyle.
-
Set short-term goals, seek support from mentors, and reward yourself for small achievements.
-
Regular exercise improves focus, reduces stress, and enhances overall mental clarity.
-
KOTC offers personalized learning tools, gamification features, and adaptive question banks to help students stay on track without burnout.