⚗️ Endothermic vs Exothermic Reaction Graphs
Understanding endothermic and exothermic reactions is a must-know concept for the MCAT, NCLEX, and general chemistry exams. These reactions don’t just test your knowledge of thermodynamics they also show up in passage-based questions, graphs, and real-world applications.
🔥 What is an Exothermic Reaction?
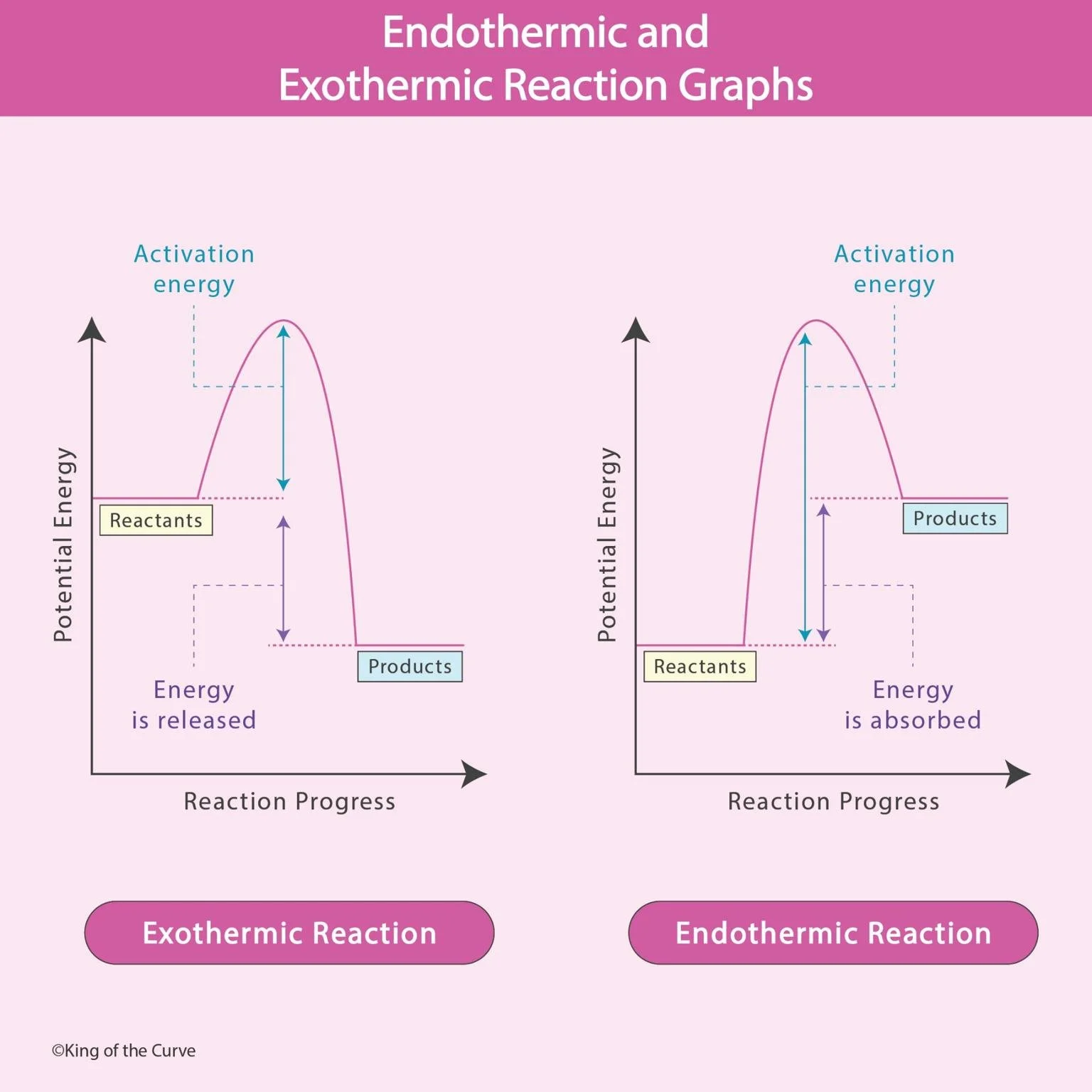
An exothermic reaction releases energy into the surroundings, usually in the form of heat.
Key Features:
Products have lower potential energy than reactants
Energy is released
ΔH (enthalpy change) is negative
💡 Examples:
Combustion reactions
Cellular respiration
💡 MCAT Tip:
If the graph shows products lower than reactants, it’s exothermic.
❄️ What is an Endothermic Reaction?
An endothermic reaction absorbs energy from the surroundings.
Key Features:
Products have higher potential energy than reactants
Energy is absorbed
ΔH is positive
💡 Examples:
Photosynthesis
Ice melting
💡 Exam Insight:
If the graph ends higher than it starts, it’s endothermic.
📊 Side-by-Side Comparison Table
| Feature | Exothermic Reaction | Endothermic Reaction |
|---|---|---|
| Energy Flow | Released | Absorbed |
| ΔH (Enthalpy) | Negative | Positive |
| Product Energy | Lower | Higher |
| Surroundings | Warmer | Cooler |
| Example | Combustion | Photosynthesis |
⚡ Activation Energy (High-Yield Concept)
Both reaction types require activation energy, which is the energy needed to start the reaction.
Represented by the peak of the graph
Even exothermic reactions need an initial energy input
Catalysts lower activation energy but don’t change ΔH
💡 MCAT Trick:
Students often confuse activation energy with total energy change—these are NOT the same.
📈 How to Read Reaction Graphs Quickly
When you see a graph like this on the exam, follow this strategy:
Look at starting vs ending energy
Identify:
Downhill → Exothermic
Uphill → Endothermic
Check the height of the peak → activation energy
👉 This method lets you answer most questions in under 10 seconds.
🧠 Real-World & Clinical Connections
These reactions aren’t just theoretical:
Metabolism → largely exothermic (energy release)
Cold packs → endothermic reactions absorbing heat
Fever responses → involve energy balance shifts
💡 NCLEX Angle:
Understanding energy flow helps explain body temperature regulation and metabolism.
🏁 Conclusion
Endothermic and exothermic reactions become simple when you focus on energy flow and graph shape. Once you master this visual pattern, you’ll be able to quickly decode even the most complex MCAT or NCLEX questions.
Frequently Asked Questions (FAQs)
-
Aim for 4-6 focused hours, ensuring you incorporate breaks to avoid burnout.
-
Practice mindfulness techniques, take practice exams under realistic conditions, and maintain a balanced lifestyle.
-
Set short-term goals, seek support from mentors, and reward yourself for small achievements.
-
Regular exercise improves focus, reduces stress, and enhances overall mental clarity.
-
KOTC offers personalized learning tools, gamification features, and adaptive question banks to help students stay on track without burnout.